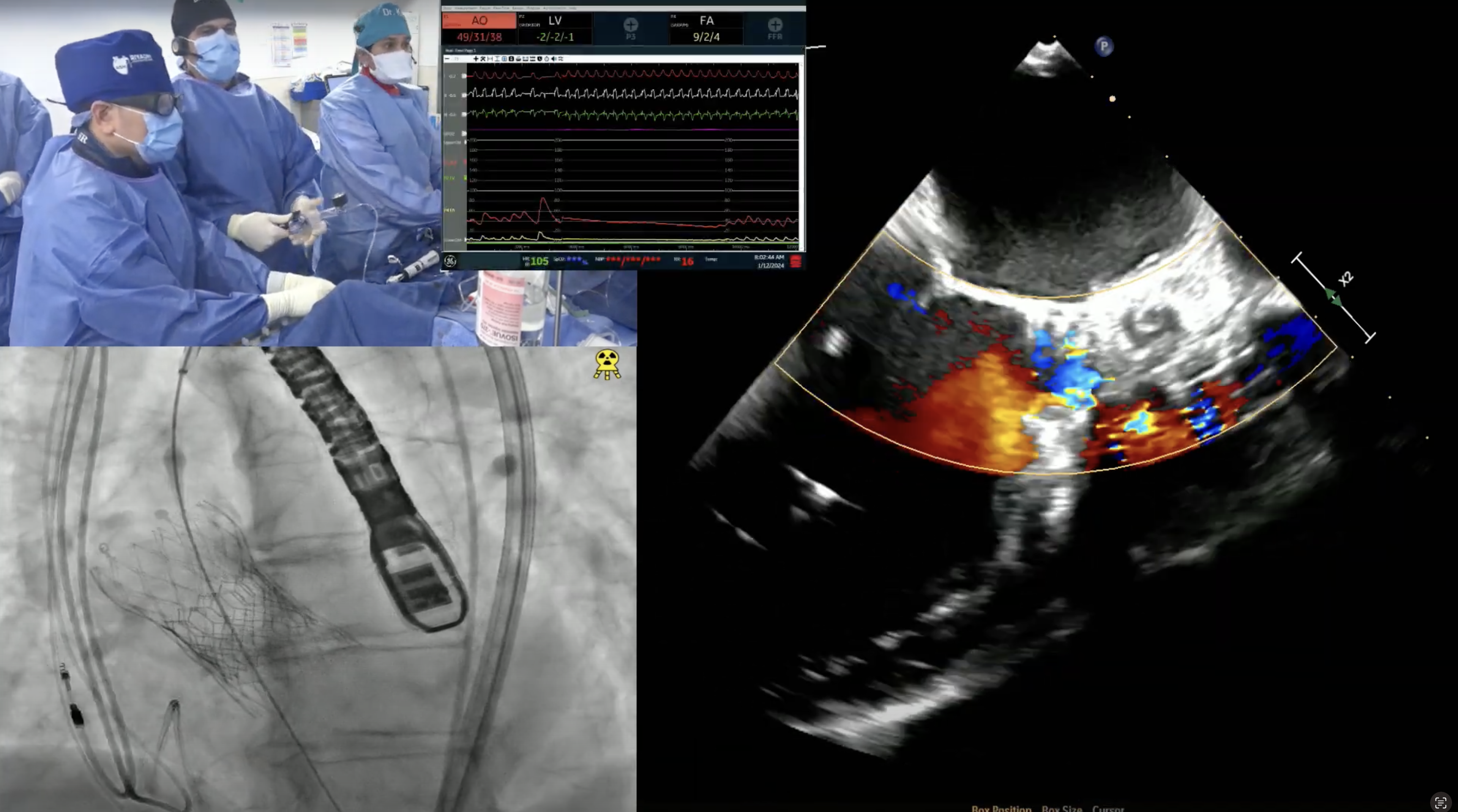
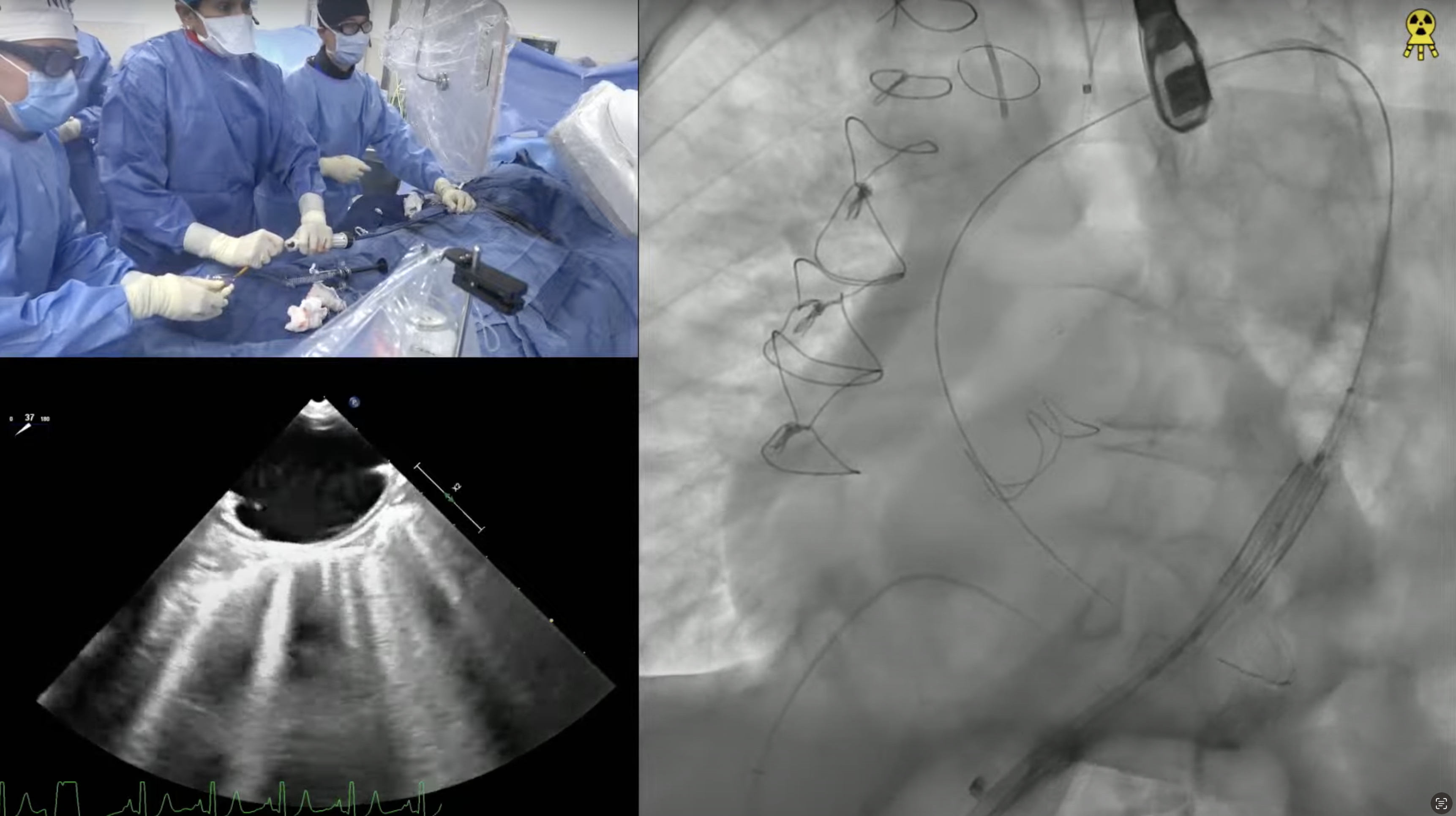
79 year old man presents with progressive exertional dyspnea (NYHA Class III) for the last 2 months. Past medical history is significant for CABG (SVG to D1 and SVG to RPL) & bioprosthetic surgical AVR (25mm Perimount 2700) in 03/09/04, atrial flutter s/p ablation, hypertension and hyperlipidemia. Recent echocardiogram revealed severe bioprosthetic aortic regurgitation due to leaflet degeneration and coaptation gap, moderate prosthetic aortic stenosis (PG/MG/AVA = 41/20/0.9) and LVEF 40-45%. Analysis of lower extremity on CT angiogram revealed adequate diameter for trans-femoral approach. The Internal Diameter of Surgical AV measured 23.1×24.6 mm (mean 23.8 mm), perimeter was 75.3mm and the area was 449.9mm2. The STS mortality risk for surgical AVR was 3.5% and logistic Euroscore mortality risk was 19.4%. The patient underwent Heart Team evaluation and was found to be at high risk for re-do SAVR due to comorbidities, advanced age and frailty. Now presents for Valve-in-Valve TAVR with 29mm Evolut-R CoreValve via left percutaneous femoral approach with Sentinel cerebral protection device.

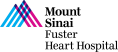

I absolutely LOVE watching these cases and share them with all of my colleagues !
I always learn so much !
Thank you Drs. Kini & Sharma ….you rock !
Fantastic case! However the the case ppt doesnt correspond to the presentation. Would be great to have Drs Kini & Sharmas ppt from the case!
regards