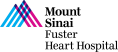Case & Plan:
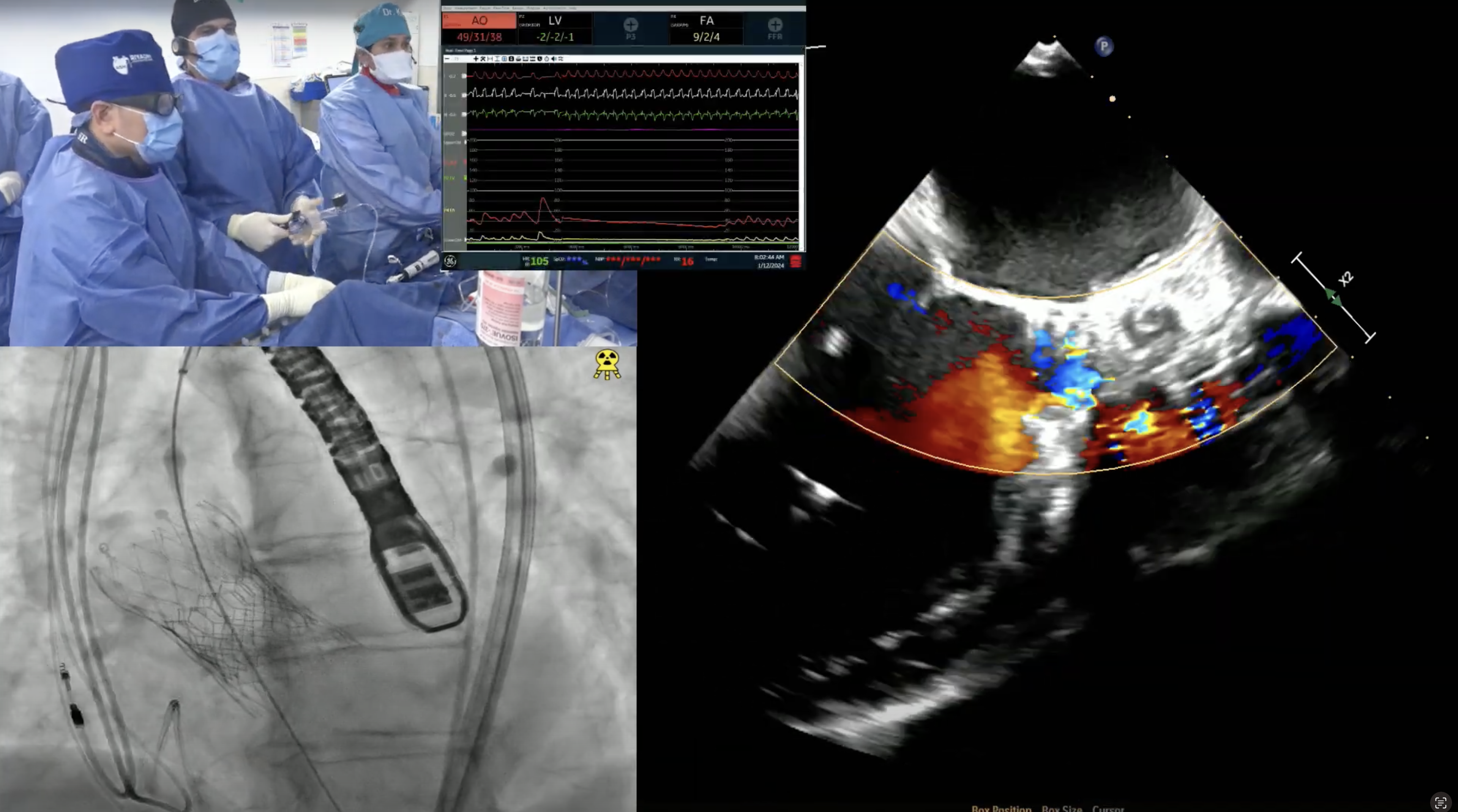
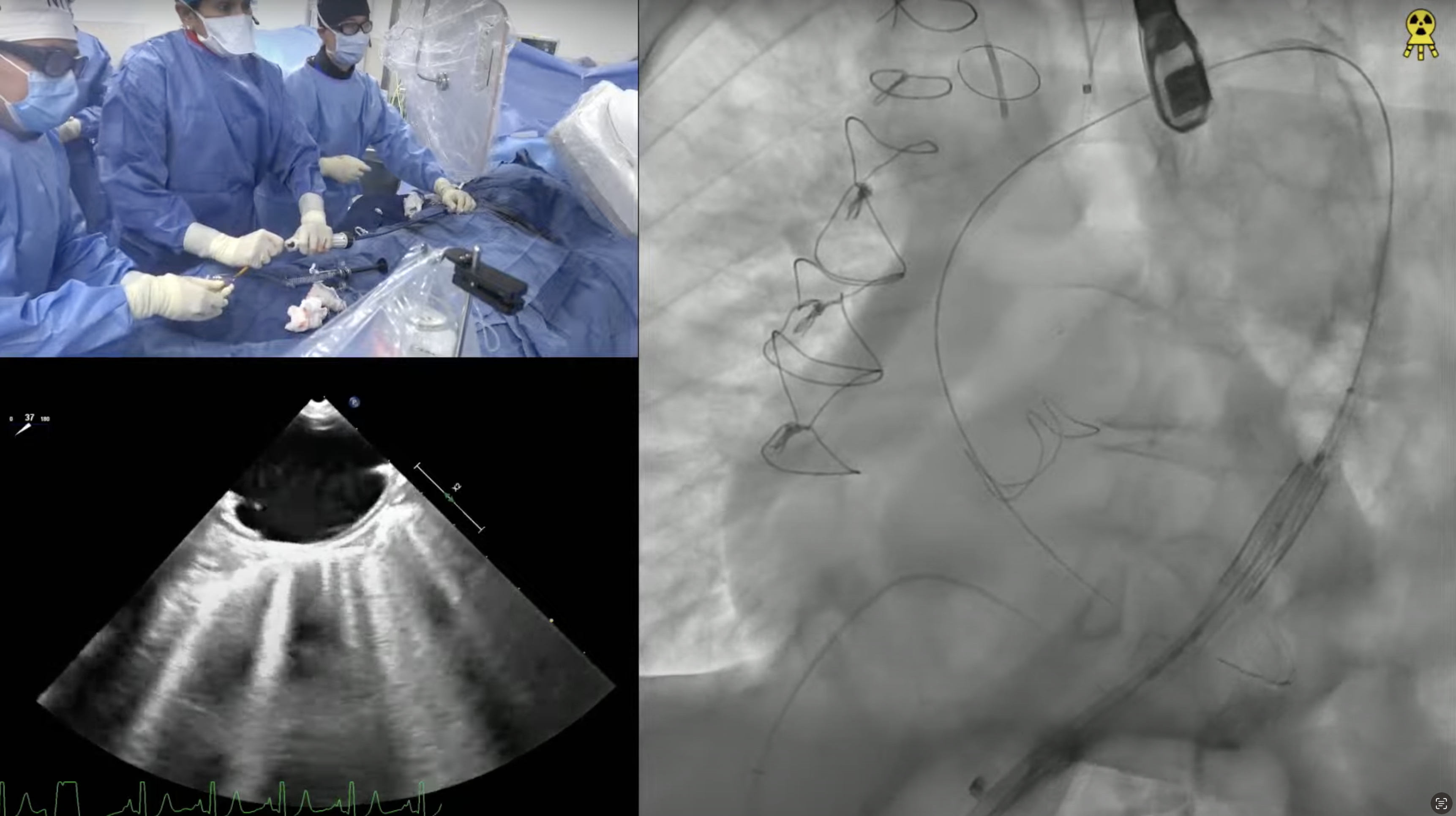
68-year-old male presents with DOE, lower extremity edema and venous stasis ulcers for 6 months. Past medical history is significant for HTN, CKD, HLD, rectal carcinoid s/p resection in 2001, carcinoid heart disease s/p bioprosthetic TVR in 2006 (33mm perimount plus 6900) and LAA excision, AF not on anticoagulation due to h/o GI bleed, alcoholic cirrhosis (Child-Pugh class B), HFrEF (now recovered) s/p BiV- ICD in 2010 with recent RV ICD lead re-positioning in to SVC (Jan 2021). Recent TEE revealed LVEF of 43% and abnormal tricuspid bioprosthesis with stenosis [mean gradient 13.3mmHg and two of the three leaflets (anterior and posterior) heavily calcified, fixed and restricted]. STS Risk for redo-TVR was calculated at 8.16%. Heart Team evaluation found him to be high risk for redo TVR due to associated medical comorbidities. Now planned for tricuspid valve-in-valve implantation (TVIV) via right percutaneous femoral venous access using a 29 (+2cc) mm SAPIEN-3 valve.